Anastomark® Coronary Graft Markers
Make Your Mark — Silicone and Stainless Steel Coronary Graft Markers
Simple Tools That Improve Surgical Confidence – Anastomark® Coronary Graft Markers
Genesee’s Anastomark® Coronary Graft Markers are available in stainless steel and silicone options, giving surgical teams flexibility based on graft location, vessel type, and long-term imaging needs. Both designs are implantable-grade and engineered to enhance post-op clarity by making bypass grafts visible under fluoroscopy, reducing contrast use, and minimizing radiation exposure. Whether placed on proximal or distal grafts, these small markers make follow-up safer, faster, and more reliable — improving outcomes for both patients and cardiologists.

Proximal Silicone Coronary Graft Markers
Anastomark® proximal silicone graft markers are made from implantable-grade radiopaque silicone and designed to conform naturally to graft anatomy. Their flexible profile and dark coloration support clear graft identification during imaging and re-operation, making them particularly useful for long-term follow-up and post-op care.
Key Highlights:
-
Implantable-grade radiopaque silicone
-
Flexible design conforms to graft anatomy
-
Dark color supports visibility during re-operation
-
Designed to support clear graft identification during follow-up imaging
Ready to learn more?
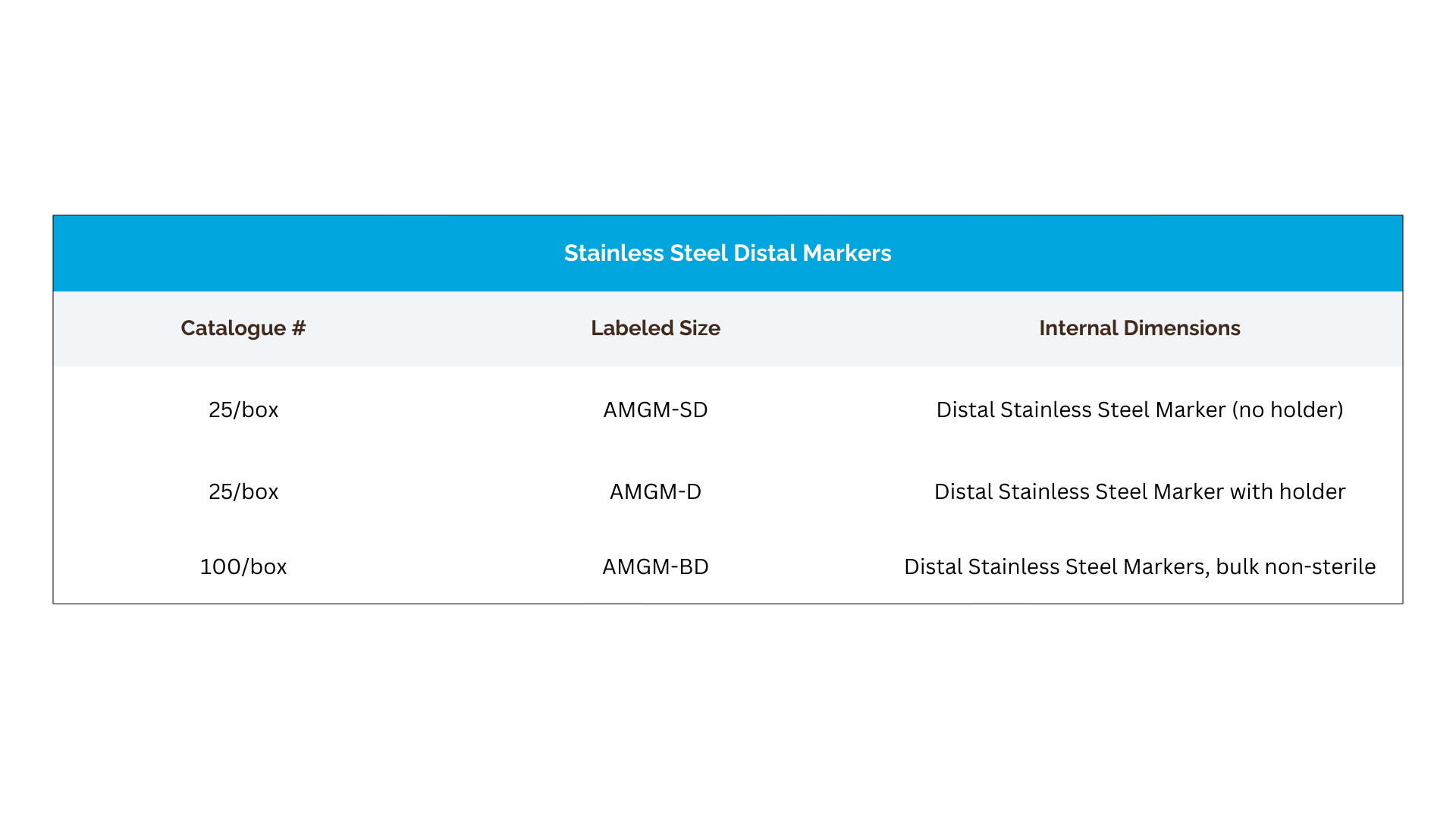
Distal Stainless Steel Coronary Graft Markers
Anastomark® distal stainless steel graft markers are made from implantable-grade stainless steel and designed to provide distinct radiographic visibility. Available with or without a holder, they support clear distal graft localization during future angiography and catheter-based follow-up.
Key Highlights:
- Implantable-grade stainless steel
- Distinct radiographic visibility
- Optional holder for easier manipulation
- Available sterile or in non-sterile bulk, single-use

Coronary Graft Marker Video
Ordering Information
Get More Information or Connect With a Local Rep
Want to learn more about our coronary graft markers or speak directly with a representative in your area? Fill out the form below and a member of our team will contact you with product details, clinical resources, or to help you schedule a consultation.
By submitting this form, you are consenting to receive marketing emails from: Genesee BioMedical Inc. You can revoke your consent to receive emails at any time by using the SafeUnsubscribe® link, found at the bottom of every email.
Explore Our Complete Cardiac Portfolio
Anastomark® Coronary Artery Bypass Graft Markers
Indications: The markers are indicated for patients undergoing coronary artery bypass graft surgery or vascular graft surgery.
Contraindications: There are no known contraindications for use.
Precautions: Take care not to damage the marker rings during handling.
MRI Conditional information can be found online at https://geneseebiomedical.com/project/graft-markers/
CAUTION: Federal law (USA) restricts this device to sale by, or on the order of, a physician.
1. Oman Z, Feghaly J, Gu L, Nasir A, Forsberg M. Utility of Radiopaque Bypass Graft Markers During Coronary Angiography
Following Coronary Artery Bypass Graft Surgery. The Journal of invasive cardiology. 2020;32(6):232-234.
2. Catheterization and cardiovascular diagnosis [Cathet Cardiovasc Diagn] 1997 Mar; Vol. 40 (3), pp. 249-53; discussion 254.
3. Pingpoh C, Siepe M, Burger K, Zietak T, Valina C, Ferenc M, Beyersdorf F, Neumann F, Hocholzer W. Impact of proximal
radiopaque coronary bypass graft markers on postbypass surgery coronary angiography. J Thorac Cardiovasc Surg 2018;155:15675-72
Design Beyond Standard
303-777-3000
800-786-4890
700 W. Mississippi Ave. #D5
Denver CO 80223 – 4509



